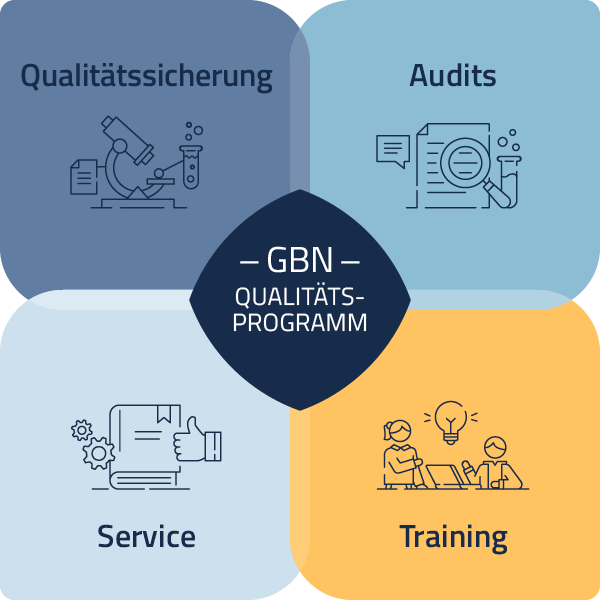
The quality of human biosamples and their associated data is of the utmost importance in order to achieve reliable and reproducible scientific results. To ensure this, all sub-processes within biobanks must be traceable and documented. A biobank therefore requires a quality management system.
In cooperation with the BioMaterialBank Heidelberg (BMBH) and the Integrated Biobank Jena (IBBJ), the GBN supports biobanks with a comprehensive quality programme. This programme is divided into four areas: quality assurance, audits, training and service.



